GIPR
March 2026
In December 2025, Eli Lilly reported positive Phase 3 clinical trial results for retatrutide, a triple-agonist peptide targeting GIPR, GLP1R, and GCGR, for the treatment of obesity. The study showed an average weight loss of 28.7% and a 75.8% reduction in WOMAC pain in patients with obesity and knee osteoarthritis after 68 weeks of treatment, underscoring retatrutide’s therapeutic potential. Additional Phase 3 trials in obesity and type 2 diabetes are underway, with results expected later in 2026.
GIPR
This month's blog post explores one of retatrutide’s key targets, glucose-dependent insulinotropic polypeptide receptor (GIPR).
GIPR is a class B1 G protein-coupled receptor involved in glucose homeostasis. Along with GLP1R and GCGR, it influences insulin release, lipid metabolism, and appetite. This makes GIPR a validated therapeutic target for type 2 diabetes and obesity, complementary to GLP-1R-based treatments.
GLP1R agonists (like semaglutide) have dominated diabetes treatment so far, but adding GIPR agonism provides complementary benefits. GIPR activation can enhance metabolic benefits (e.g., insulin release and improved lipid metabolism) while potentially mitigating gastrointestinal side effects associated with GLP1R-only therapy. Dual (GLP1R/GIPR) or triple agonists (GLP1R/GIPR/GCGR) are emerging as valuable treatments, showing high efficacy with fewer side effects.
Retatrutide is a prime example of a triple agonist, and a recent structural study has revealed how this peptide binds to each of the three target receptors, elucidating the structural basis of its therapeutic action.
Structural Basis of Retatrutide's Triple Agonism
Recent cryo-EM structures of the peptide retatrutide bound to GIPR, GLP1R, and GCGR unravel the molecular basis for its triple agonism. Key findings include:
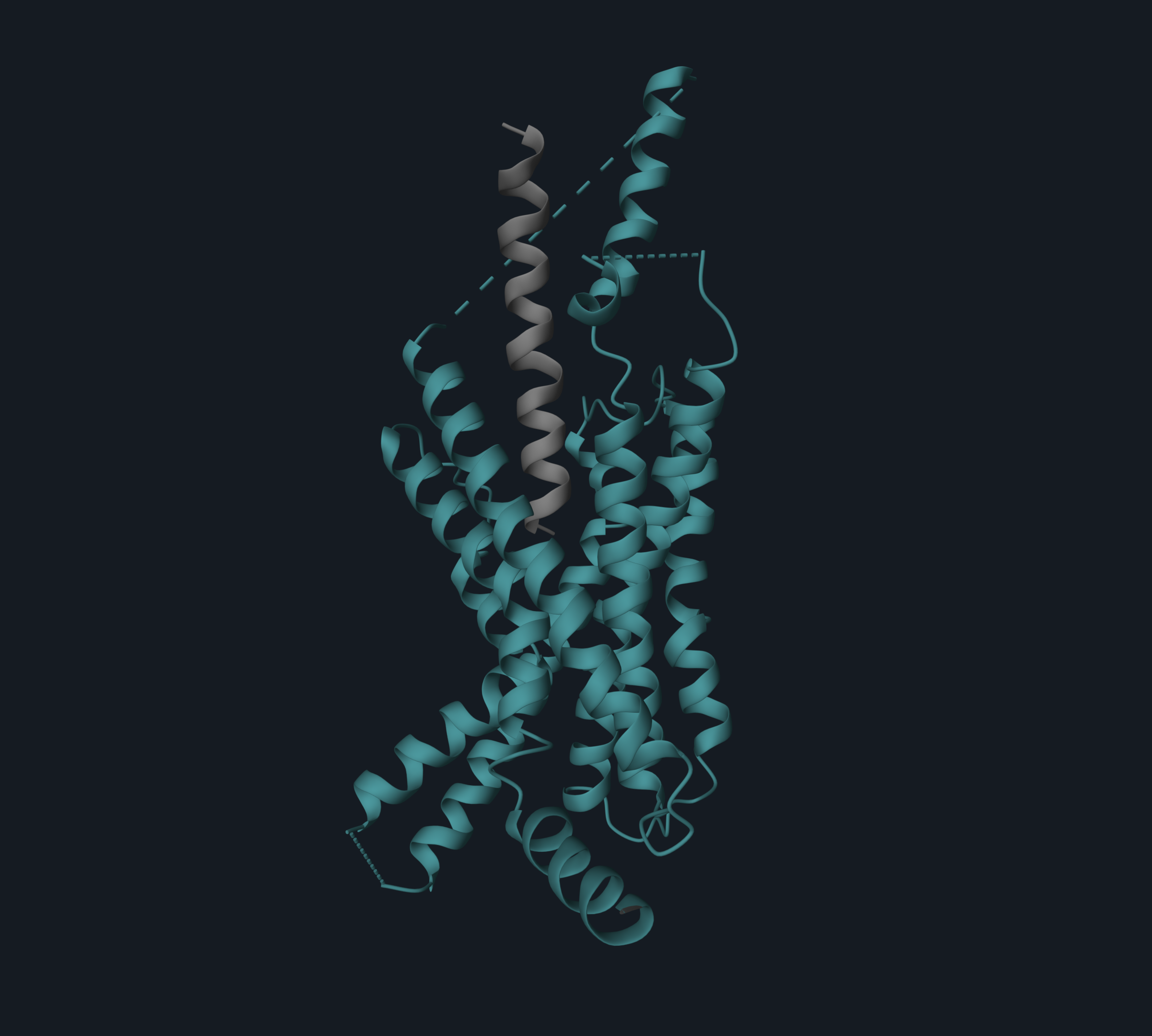
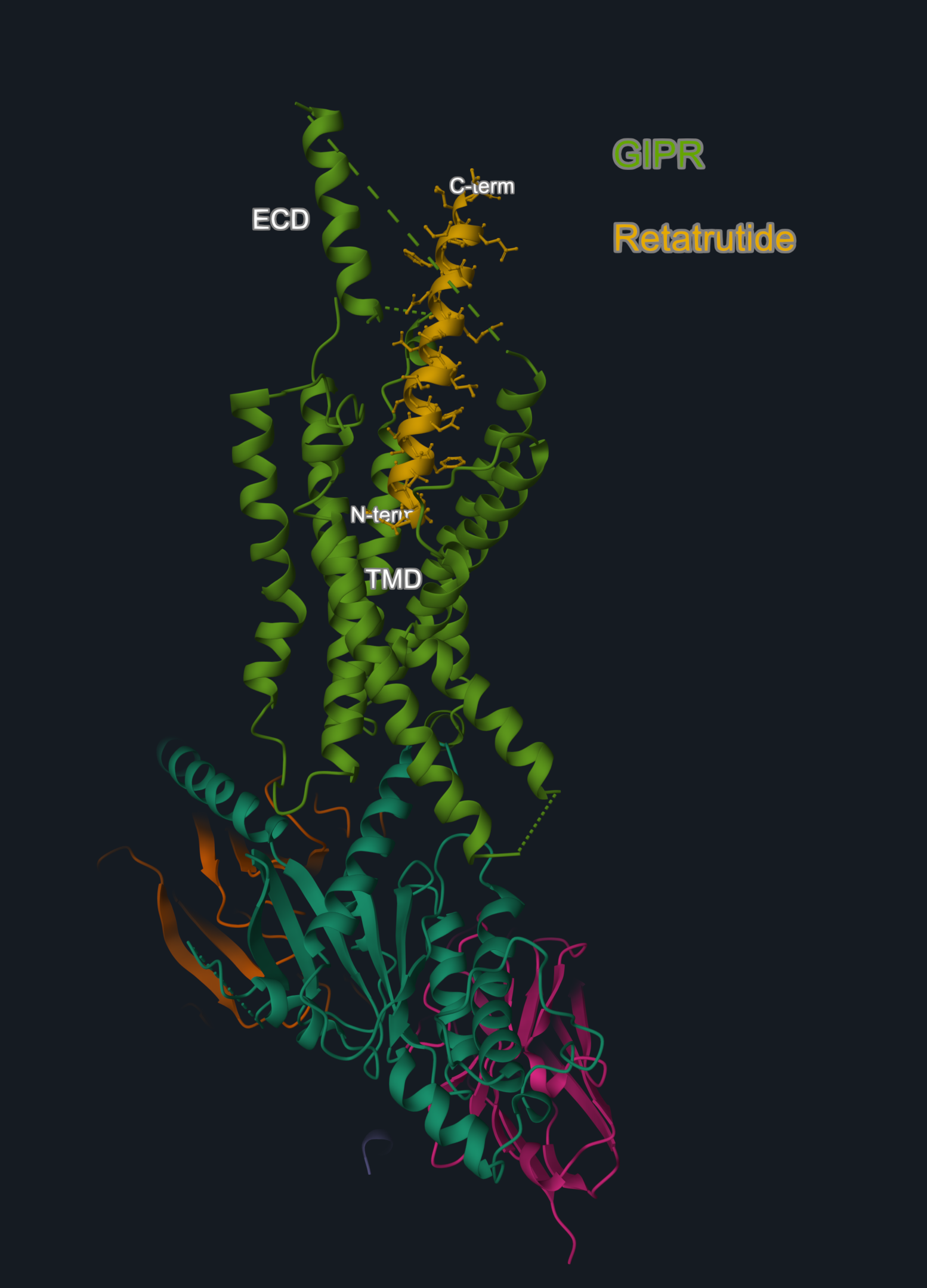
Retatrutide adopts a single continuous helix that inserts into the transmembrane domain (TMD) of the receptors via its N-terminal segment (residues 1–13), while its C-terminal (residues 14–30) interacts with the extracellular domain (ECD).
Image 1. Cryo-EM structure (PDB code: 8YW4) of GIPR (in light green) in complex with peptide retatrutide (in yellow). The N-terminal of the peptide is buried in the receptor transmembrane domain (TMD), and the C-terminal engages the extracellular domain (ECD).
Image is produced with the 3decision® software.
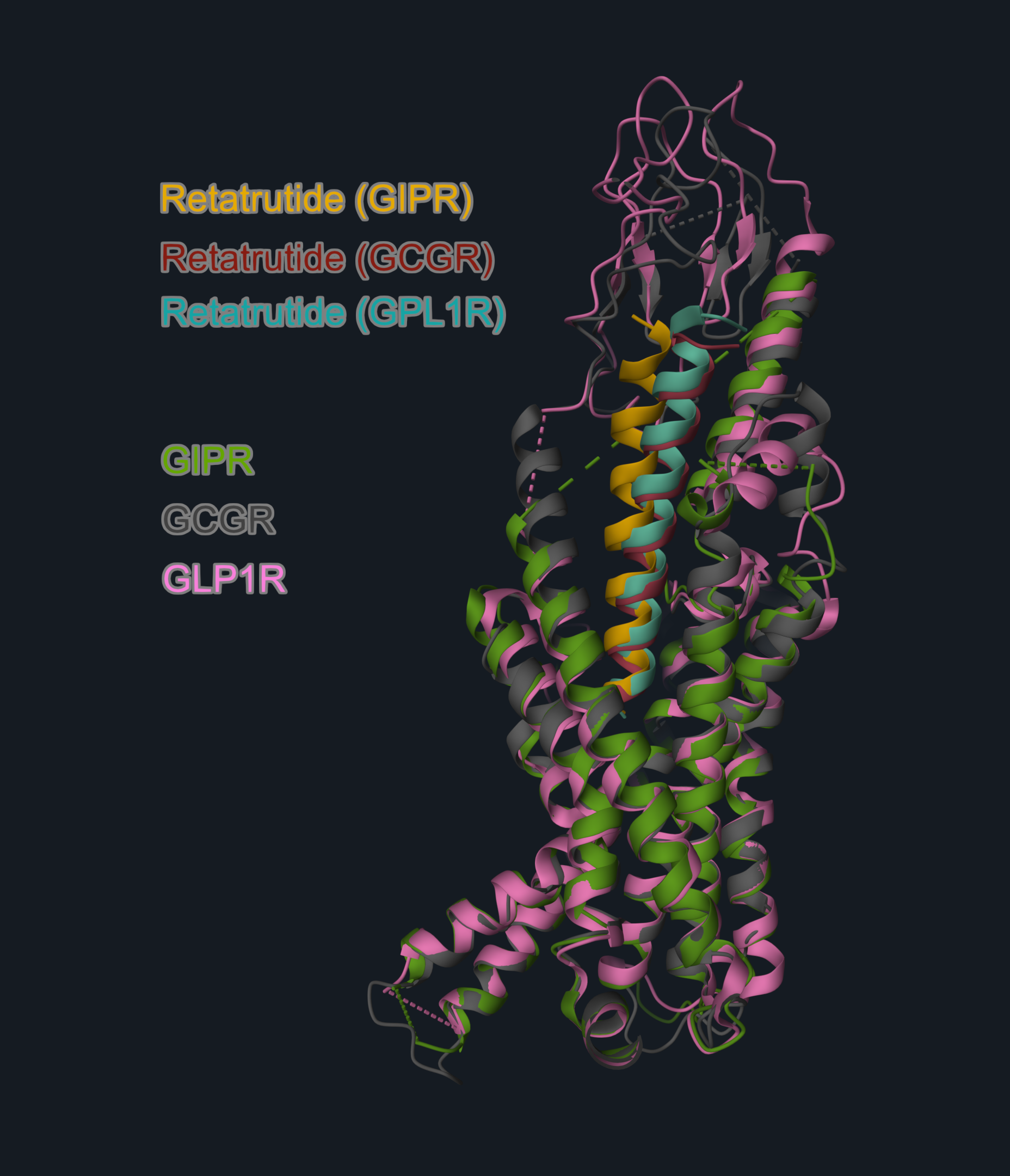
The overall structures of retatrutide-bound receptors are highly similar, but each receptor has a few unique interactions with the peptide. These subtle differences translate into varying potency at each target: retatrutide is a super-agonist at GIPR (8.9 times more potent than the native GIP peptide) and is less potent at GLP-1R and GCGR. Still, the combination of common and unique contacts enables this single molecule to activate all three receptors effectively.
Image 2. Cryo-EM structures of retatrutide in complex with: GIPR (PDB code: 8YW4, receptor in green, peptide in yellow); GCGR (PDB code: 8YW5, receptor in grey, peptide in red); GLP1R (PDB code: 8YW3, receptor in pink, peptide in teal). Structures are aligned on the receptor chain (reference structure: GIPR: retatrutide complex, GIPR chain). The receptors are aligned on the GIPR structure to highlight the similar overall binding arrangement.
Overlay and image generated with the 3decision® software.
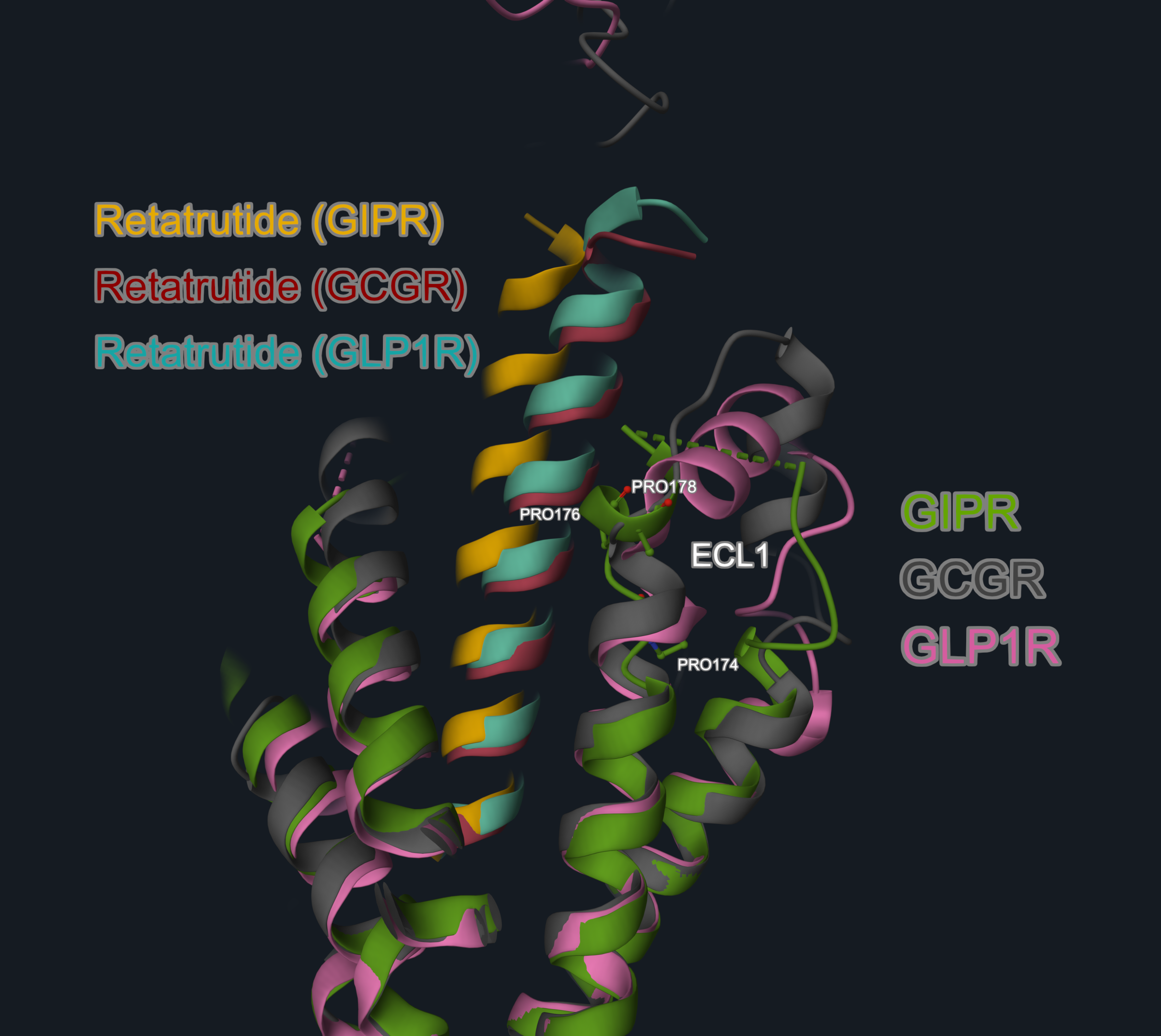
A key differentiator on the retatrutide binding mode is at the level of the extracellular loop-1 (ECL1). In GLP1R and GCGR, ECL1 forms a short helix. In GIPR, this same region adopts an unwound and relaxed loop conformation. This flexibility is likely due to the presence of three proline residues in GIPR’s ECL1 (P194, P196, P198). This allows the peptide to straighten and shift its N-terminal tip towards the GIPR transmembrane core by ~4 Å, compared to GLP-1R or GCGR. This conformational plasticity enables GIPR to accommodate a broader range of peptide sequences relative to GLP1R and GCGR, which may partly underlie the enhanced potency of retatrutide at GIPR.
Image 3. Focus on the ECL1 region of the retatrutide complexes (GIPR complex, PDB code 8YW4, receptor in green, peptide in yellow; GCGR complex, PDB code 8YW5, receptor in grey, peptide in red; GLP1R, PDB code 8YW3, receptor in pink, peptide in teal). The proline residues in GIPR ECL1 region are in the balls-and-stick representation and labeled.
The structures overlay and image are produced with the 3decision® software.
From a peptide design perspective, this structural insight implies that GLP-1R and GCGR demand specific amino acids at positions in contact with the ECL1 to accommodate the ECL1’s rigid structure and maintain optimal binding, whereas GIPR tolerates greater sequence variability in this region due to the flexibility of its ECL1, opening up opportunities for further structure-informed peptide optimization.
Conclusion
The structural characterization of retatrutide’s interactions with GIPR, GLP-1R, and GCGR offers an example of how detailed molecular insights can drive therapeutic innovation.
By understanding exactly how one peptide engages multiple receptors, researchers can better design the next generation of drugs, with improved efficacy, selectivity, and safety in the treatment of complex metabolic diseases.
Reference:
Li, Wenzhuo et al. “Structural insights into the triple agonism at GLP-1R, GIPR and GCGR manifested by retatrutide.” Cell discovery vol. 10,1 77. 17 Jul. 2024, doi:10.1038/s41421-024-00700-0